
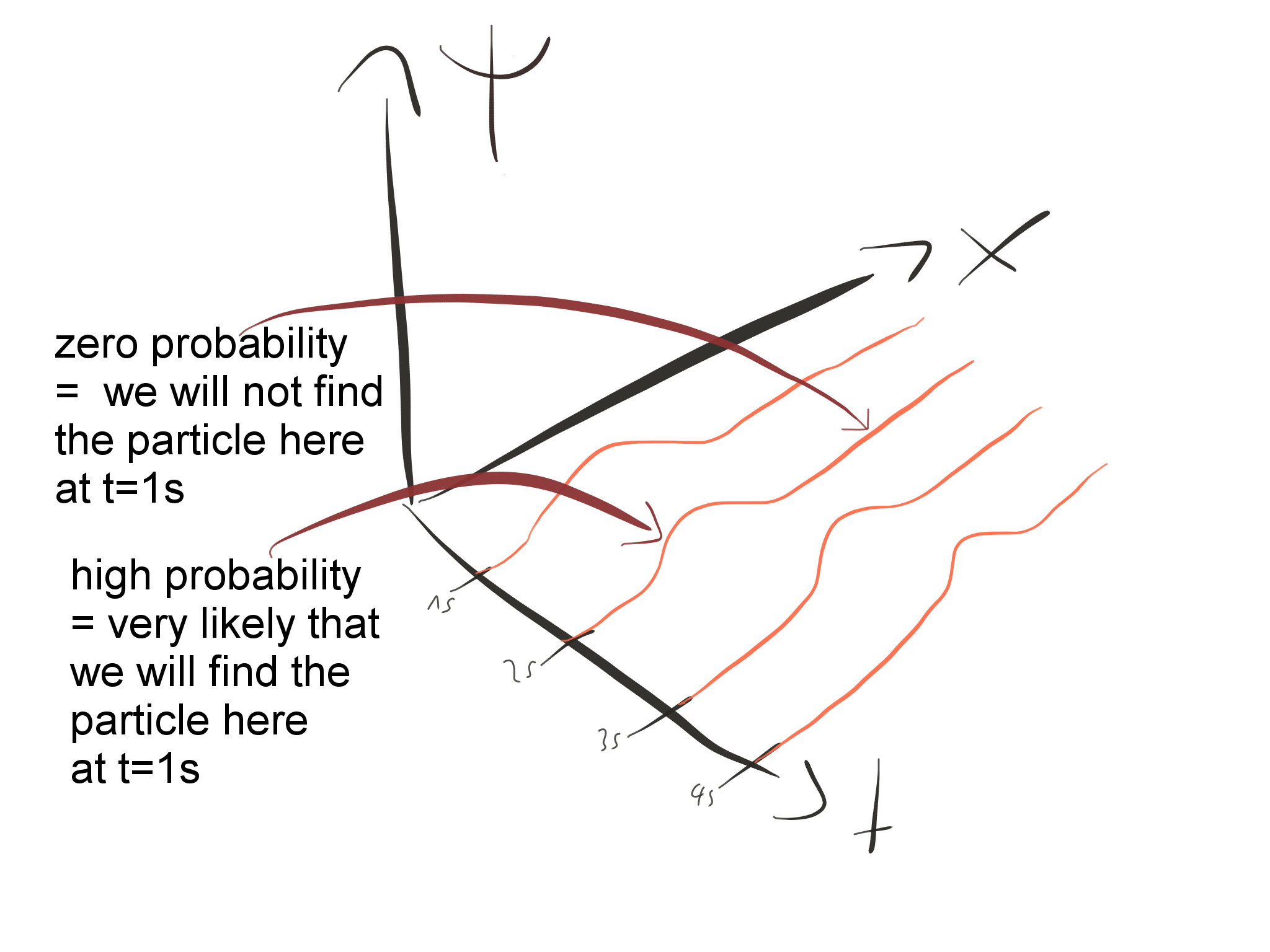
For example, with our particle in a box gives the probability density for finding the particle at position But it is also possible to solve Schrödinger’s equation for many particle systems and to find wave functions for other observable quantities, for example the momenta of the particles. In quantum mechanics the information about the system is contained in the solution to Schrödinger’s equation, a wave function The square of the absolute value of the wave function, is interpreted as a probability density.
#Schrodinger equation explained full
In classical mechanics what you’re after are the positions and momenta of all particles at every time : that gives you a full description of the system. He received the Nobel Prize in Physics in 1933 for having managed to develop the Schrödinger equation and after maintaining a long working relationship with Albert Einstein, he proposed the thought experiment of Schrödinger’s cat in which he showed the paradoxes and questions to which quantum physics was involved.Schrödinger’s equation is to quantum mechanics what Newton’s second law of motion is to classical mechanics: it describes how a physical system, say a bunch of particles subject to certain forces, will change over time. Schrodinger made important contributions in the field of quantum mechanics and thermodynamics. These are areas where the probability of finding an electron is considerably higher. The referred probability is proportional to the square of the wave function Ψ2. The postulates of the atomic model are the following:Įlectrons behave like standing waves that are distributed in space according to the wave function Ψ.Įlectrons move inside the atom in orbitals.
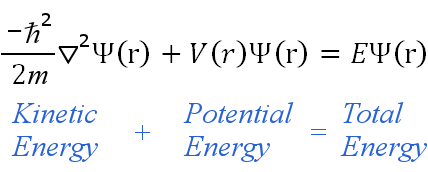
Postulates of the atomic model of Schrodinger
